Abstract
Introduction
Vascular endothelial injury related to treatment with proteasome inhibitors (PI) has been previously described. Carfilzomib is an irreversible PI and has been associated with cardiovascular toxicity, suggesting increased risk of endothelial injury. Thrombotic microangiopathy (TMA) has been described in multiple myeloma (MM) patients receiving PIs; more often with carfilzomib. In the pediatric transplant population, increased risk of TMA was related to heterozygous mutation in the alternative complement pathway. The homozygous deletions enable uncontrolled complement activation and was found three times more frequently in TMA population. We hypothesized that MM patients with complement mutation are at increased risk of PI-related TMA.
Materials and Methods
We identified ten cases of renal TMA in MM patients receiving carfilzomib from two medical institutions in Greece and in the United States. TMA was diagnosed based on either renal biopsy or acute kidney injury, new-onset anemia, thrombocytopenia and increased LDH in the absence of disease progression. We performed targeted sequencing of the twelve genes implicated in TMA: CFH, CFI, MCP, CFB, CFHR5, C3, THBD, DGKE, PLG, ADAMTS13, MMACHC and G6PD genes. Multiplex Ligation-Dependent Probe Amplification (MLPA) was performed for the analysis of the CFH-CFHR5 region.
Results
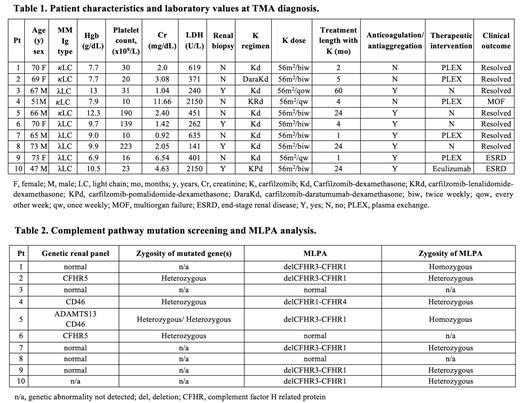
Patient characteristics and laboratory values at TMA diagnosis are presented in Table 1. The median age of patients was 68 years (range, 47-73), with slight male predominance (60%). Median laboratory values at diagnosis included hemoglobin 9.35 g/dL, platelet count 26,500 x 10 6/L, LDH 356,5 U/L and creatinine 2.25 mg/dL. Patients were treated with carfilzomib doses ranging 20-70 mg/m 2. Regimens included carfilzomib-dexamethasone (Kd, 7 patients), carfilzomib-lenalidomide-dexamethasone (KRd, 1 patient), carfilzomib-pomalidomide-dexamethasone (KPd, 1 patient), and carfilzomib-daratumumab-dexamethasone (DaraKd, 1 patient). All patients had previously received at least one line of therapy and seven patients had previously undergone autologous stem cell transplant (ASCT), 1-12 years prior to TMA diagnosis. The median time between carfilzomib initiation and TMA diagnosis was 4.5 months (range, 1-60 months). Diagnosis was confirmed with renal biopsy in four cases. Carfilzomib was discontinued in all patients and five patients were treated with plasma exchange (PLEX) while one patient received eculizumab. Seven patients demonstrated clinical improvement and resolution of TMA at 1 year after discontinuation of carfilzomib. Two patients progressed to end stage renal disease (ESRD) requiring intermittent hemodialysis, and one patient developed multiorgan failure. Results of genetic panel are shown in Table 2. Deletions of the CFHR3-CFHR5 region were present in seven cases (70%): two patients carrying a homozygous deletion of CFHR3-CFHR1, four patients with a heterozygous deletion of CFHR3-CFHR1, and one patient with heterozygous deletion of CFHR1-CFHR4. Direct gene sequencing revealed identifiable mutations in CD46 (MCP) and CFHR5 in two distinct patients. The functional correlation and clinical significance are yet to be investigated.
Conclusions
In our cohort of ten patients of carfilzomib-induced TMA, deletions of CFHR3-CFHR5 occurred frequently (70%). In the setting of carfilzomib use, heterozygous CFHR3-CFHR1 deletion may represent a risk factor for the development of TMA. Our data set the bases for larger studies assessing complement mutation as a predisposing factor for PI-induced TMA.
Kastritis: Amgen: Consultancy, Honoraria, Research Funding; Genesis Pharma: Honoraria; Janssen: Consultancy, Honoraria, Research Funding; Takeda: Honoraria; Pfizer: Consultancy, Honoraria, Research Funding. Dimopoulos: Janssen: Honoraria; BMS: Honoraria; Takeda: Honoraria; Beigene: Honoraria; Amgen: Honoraria. Richardson: AbbVie: Consultancy; Secura Bio: Consultancy; Sanofi: Consultancy; GlaxoSmithKline: Consultancy; Karyopharm: Consultancy, Research Funding; Regeneron: Consultancy; AstraZeneca: Consultancy; Oncopeptides: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Janssen: Consultancy; Celgene/BMS: Consultancy, Research Funding; Protocol Intelligence: Consultancy; Jazz Pharmaceuticals: Consultancy, Research Funding. Bianchi: Jacob D. Fuchsberg Law Firm: Consultancy; MJH: Honoraria; Karyopharm: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal